What is Biomedical Research
Biomedical scientists study human physiology and the treatment or understanding of disease. Biomedical research applies the principles of the physical sciences to medicine. Most biomedical research is conducted by physicians or biomedical scientists, but many studies are conducted by biologists, chemists, physicists, and other medical and scientific professionals.
Most biomedical research involves clinical trials, which are phased studies using human volunteers, designed to answer safety and efficacy questions about biologics, devices, pharmaceuticals, new therapies or new ways of using known treatments. Trials are often conducted in small group initially but expanding in later stages once safety and efficacy are demonstrated. Most clinical trials are FDA regulated, but there are some exceptions.
Types and Methods
- Studies designed to evaluate the safety, effectiveness, or usefulness of an intervention:
- research on therapies (e.g., drugs, exercise, surgical interventions, or medical devices)
- diagnostic procedures (e.g., CAT scans, prenatal diagnosis through amniocentesis)
- preventive measures (e.g., vaccines, diet, or fluoridated toothpaste)
- Research on normal human functioning and development:
- studies of the human body while exercising, fasting, feeding, sleeping, or learning
- responding to such things as stress or sensory stimulation
- Studies comparing the functioning of a particular physiological system at different stages of development (e.g., infancy, childhood, adolescence, adulthood, or old age)
- Studies defining normal childhood development so that deviations from normal can be identified
- Records research – often used to develop and refine hypotheses
- Research on specific disease processes is often needed before improved methods of prevention, diagnoses, and treatment can be developed, for example:
- research on the biochemical changes associated with AIDS
- research on the neurological changes associated with senile dementia
- Research on the human genome and genetic markers – for the purpose of creating new avenues for understanding disease processes and their eventual control
- Other biomedical studies that do not involve human subjects or are exempt from the human subjects regulations, and, therefore, do not require IRB review:
- research with animals
- research on preexisting samples of materials (tissue, blood, or urine) collected for other purposes, where the information is recorded by the investigator in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects
- research based on records, when the data are recorded in such a manner that the individuals to whom the records pertain cannot be identified, either directly or through identifiers linked to them
Risks/Harm
Risk is the probability of harm or injury (physical, psychological, social, or economic) occurring as the result of participation in a research study. Biomedical researchers must consider the following risks when conducting their study:
- Social, psychological, or economic harm (See Social Behavioral Research for details)
- Physical harm
- exercise-induced or repetition-exacerbated physical harm, such as carpal tunnel syndrome, stress fractures, asthma attacks, or heart attacks
- exposure to minor pain, discomfort (e.g. dizziness), or injury from invasive medical procedures
- possible side effects of drugs
Although most of the adverse effects that result from medical procedures or drugs are temporary, investigators must be aware of the potential for harm. The IRB will want to know how such outcomes will be minimized or addressed and is responsible for conducting a risk/benefit assessment.
iStar
When submitting an application in iStar you will see the following in section 1.5.
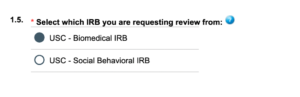
Hover over the ![]() for a definition of USC Behavioral IRB:
for a definition of USC Behavioral IRB:

Genomic Data Sharing (GDS)/Stem Cell Research
- USC Stem Cell Regenerative Medicine Initiative
- USC Stem Cell Research Oversight Committee (SCRO)
- NIH Genomic Data Sharing Policy (formerly ‘GWAS’)
- California Institute for Regenerative Medicine (CIRM)
- Genetics and Public Policy Center
- NIH: Stem Cell Research
- U.K. Human Fertilisation and Embryological Authority
- The Hinxton Group: An International Consortium on Stem Cells, Ethics and Law
- The International Society for Stem Cell Research (ISSCR)
- Human Genome Project: Educational Kit

